|
12/30/2023 0 Comments Calcium chloride molar mass 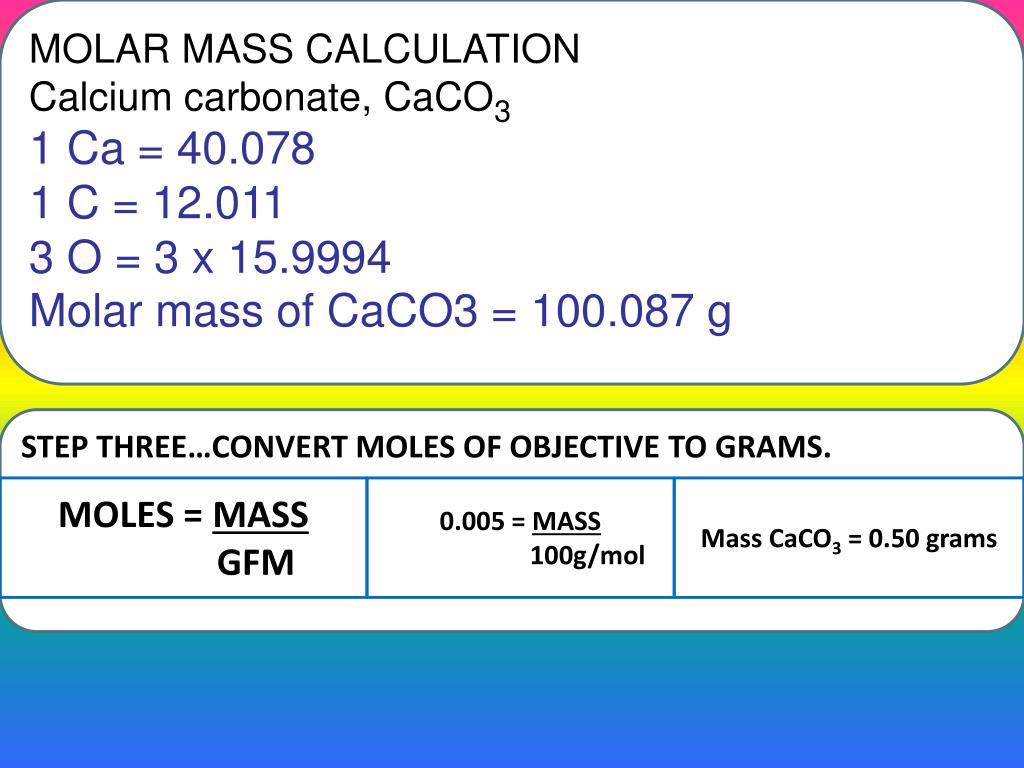
In Imperial or US customary measurement system, the density is equal to 114.243 pound per cubic foot lb/ft, or 1.058 ounce per cubic inch oz/inch. density of calcium chloride tetrahydrate is equal to 1 830 kg/m. Percent purity can be calculated using: \[\% \text\%\) if your sample is not completely dry.) Conclusionīy performing an experiment we were able to calculate the percentage yield of a reaction. Calcium chloride tetrahydrate weighs 1.83 gram per cubic centimeter or 1 830 kilogram per cubic meter, i.e. Or you may need to know how much of a particular ion is dissolved in water to determine if it is below the legally allowed level. Molar mass of K 39.1 g Molar mass of Mn 54.9 g Molar mass of O 16.0 g Molar mass of KMnO4 39.1 g + 54.9 g + (16.0 g x 4) Molar mass of KMnO4 158. General description Calcium chloride dihydrate is a moisture resistant, cheap and commonly available calcium salt. Percent purity is important since when you make a compound you may have a small amount of impurity in the sample and you would need to keep this below a certain level. The molecular mass of this drug is Ca 1×40 40 Cl 2×35.5 71 CaCl2. Molecular weight calculation: 40.078 + 35. Convert grams CaCl2 to moles or moles CaCl2 to grams. The final use of stoichiometric calculations that we will look at is to determine the percent purity of a sample. There are 1 calcium atom and 2 chlorine atoms in this calcium chloride structure. This compound is also known as Calcium Chloride. Optional Experiment: The thermal decomposition of lead(II) nitrate.Optional Video: TD of Lead Nitrate Video Actual Question: What mass of calcium chloride, CaCl2 (molar mass 111.0 g/mol) is needed to prepare 2.850 L of a 1. The molecular weight of Calcium Chloride (CaCl2) is 110.9834.2) Divide 2.97 g by the molar mass of CaCl22H20 (aq) you got in 1). Meanwhile, one carbon-12 atom has a mass of exactly 12 amu, i.e. We know that the mass of one mole of carbon-12 atoms is 12 g, or, 1 molm12C12 g, because of the definition of the mole. We now need to convert from amu to g/mol. Step 3: Calculate the mass of calcium carbonate 1) Simply use the molar masses provided by the periodic table to calculate the molar mass. it shows us that calcium has a mass of around 40 amu.Supplier details: American Elements 10884 Weyburn Ave. CALCIUM CHLORIDE, HYDRATED Chemwatch: 31224 Version No: 5.1.1.1 Safety Data Sheet according to WHS and ADG requirements Issue Date: Print Date: S. Relevant identified uses of the substance: Scientific research and development. KALSYUM KLORR Chebi 3312 RTECS number EV9800000 ATC code A12AA07, B05XA07, G04BA03 Molecular formula CaCl2 Molar mass 110.98 g / mol (anhydrous). The molar mass of chlorine is 35.5 grams per mole. 77.5 grams This is Expert Verified Answer 47 people found it helpful ChoiSungHyun Explanation: Molar mass of CaCl2 110.98g/mol Moles of CaCl2 30.0 / 110.98 0. Step 2: Calculate the number of moles of calcium carbonate Product Number: All applicable American Elements product codes, e.g. How many grams of CaCl2 contain 15.0 grams of chlorine Give your answer to one decimal place. (Molar mass of Ca 40.078 g/mol, Cl 35.453 g/mol, O 15.999 g/mol, Ag 107.868 g/mol, N 14.007 g/mol) A.Step 1: Calculate the number of moles of calcium chloride.Step 1: Write down an equation for percent purity. 
\): Calcium chloride is used as a drying agent and as a road deicer.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
